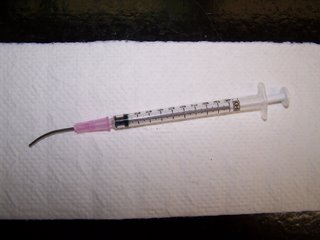
Mice are infected by gavage (per os) using a blunted tip needle (18 g) that can be commercially obtained or you can make your own. To make your own gavage needle, completely blunt the tip of an 18 g needle using a file or grinder. Be sure the needle is completely polished and does not contain any burrs that could lacerate the mouse's esophogus. Carefully bend the needle into a very shallow curve (a picture of a 'homemade' needle will be posted).
To achieve the appropriate larval concentration, perform the following:
1. Place the larval suspension in a small beaker (10-30ml) containing a small magnetic stir bar.
2. With continual mixing withdraw an aliquot (50 - 100 ul) using the 1 ml syringe and gavage needle that will be used to infect the mice.
NOTE: The stir bar must rotate with sufficient speed to effect thorough mixing but not so fast as to create centripetal forces which concentrate the larvae in a vortex. It is also important the dosing syringe is always placed at the same location in the beaker and at the same angle when withdrawing aliquots for larval counts.
3. Adjust volume of larval suspension with distilled water to approximate final concentration (x no. of larvae/0.2 ml).
4. Make 5 counts. Counts should not vary more than 10% above or below the mean.
5. Mice are typically infected with 100-150 larvae (source mice with 200 larvae). Work quickly after filling syringe with larvae as they settle very fast. The major source of variability in infecting mice is INCONSISTENCY.
6. Depending on the mouse strain, about 70-80% of the innoculated larvae will survive to adulthood.
7. For egg source mice infect male, thy1.1 BALB/C mice with 200 larvae. You will get good egg production for about 2 months.

4 comments:
Thanks a lot for all the helpful information on your blog. I would like to know if you anesthetize the mice prior to infection.
Thanks,
Sandra
Hi Sandra. It is not necessary to anesthetize the mice when infecting them. It is a very quick process that doesn't seem to cause the mice much discomfort if you use the proper size gavage needle.
Another question: Are you really using 0.2mlto infect the mice? Mine will not swallow such a large volume :-(
Thanks.
Sandra
There should be no swallowing involved if the mice are intubated properly. 0.2 ml is not a large volume for even a 12 gram mouse.
Post a Comment